Gap junctions are specialized membrane structures consisting of intercellular channels that connect adjacent cells in many tissues and organs. These structures provide chemical and electrical communication. They are assembled from connexin proteins. One protein, connexin 43 (Cx43) is universally expressed and frequently studied, and its role in cardiovascular development and function is one focus of cellular biologist Matthias Falk.
Falk, professor of biological sciences, and his team have long studied cells in culture, and are now collaborating with Kathy Iovine, professor of biological sciences, who studies development using the zebrafish model. Zebrafish also have Cx43 and the protein functions similarly to humans. The parallel is that connexin mutations also cause a variety of human diseases.
“We found these developmental defects in zebrafish; mainly, in every organ where Cx43 is expressed in a large amount, and there's quite a number of organs that show defects,” Falk says. “The most prominent that was quite easily detectable was heart and vasculature.”
Connexins have an unusually short half-life of only one to five hours, resulting in constant endocytosis and biosynthetic replacement of gap junctions, which still puzzles researchers. The Cx43 Carboxy-terminal domain serves as the regulatory hub of the protein affecting all aspects of gap junction function. The cell modifies this domain on specific amino acids, either by phosphorylation, by adding ubiquitin moieties, or by binding and releasing scaffolding proteins to close the channels and allow them to interact with the endocytic machinery. Cells use the endocytic adaptor protein AP-2 in conjunction with clathrin and other mechanisms to create pathways to internalize gap junctions.
Falk and his colleagues, including former Ph.D. student Caitlin Hyland, study how the loss of a specific C-terminal region known to regulate gap junction turnover, effects gap junction function. They found that the C-terminal deletion causes defective gap junction endocytosis resulting in increased gap junction intercellular communication. Increased Cx43 protein content in zebrafish, specifically in the cardiac tissue, larger gap junction plaques and longer Cx43 protein half-live coincides with severely impaired development.
In the lab, Cx43 zebrafish show severe defects of the cardiovascular system, including malformed, elongated hearts, decreased heart rate, malformed, unorganized vasculature, and impaired blood flow, indicating that undisturbed gap junction endocytosis is crucial for normal organ development. The team has demonstrated for the first time that Cx43 gap junction endocytosis is an essential aspect of gap junction function and when impaired, gives rise to significant pathologies including cardiovascular defects.
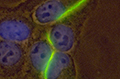
Image above: A fluorescence light microscopic image of gap junctions consisting of thousands of gap junction channels that Falk visualized in human epithelial cells by tagging the gap junction protein connexin 43 with green fluorescent protein (GFP, green). The cell nucleus is visualized in addition with DAPI (blue).






